Selected Reviews of AAO 2011 Retina SubSpecialty Day Presentations
Here is another overview of a presentation made during the Retina SubSpecialty Day Meeting.
Dr. Elias Reichel, of Tufts University School of Medicine and a founder of Hemera Biosciences, Inc., of Boston, MA, presented on a new approach to treating the dry form of age-related macula degeneration. His paper was based on the research being done by Hemera Biosciences on HMR59, a naturally occurring protein that protects retinal cells from damage by MAC (Membrane Attack Complex), that can be delivered for long-lasting activity via a gene therapy approach.
HMR59 was developed at Tufts University and subsequently licensed to Hermera Biosciences.
Complement Regulation via Gene Therapy for Dry AMD
Elias Reichel, M.D., Professor of Ophthalmology, Tufts University School of Medicine, Boston, MA
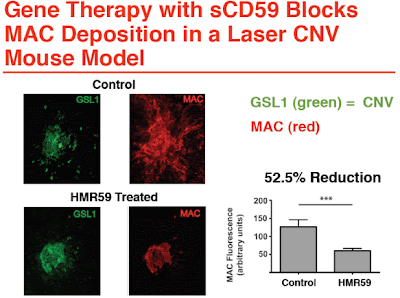
HMR59 is a novel therapy primarily targeted at geographic atrophy and other forms of dry age related macular degeneration (Dry AMD) by blocking the final stage of the complement cascade, membrane attack complex (MAC). The complement cascade is implicated via genetic studies as playing a critical role in both wet and dry AMD.
HMR59 is a gene therapy using an AAV2 vector to express a soluble form of a naturally occurring membrane bound protein called CD59 (sCD59), which blocks MAC. Membrane attack complex is the final common pathway of activation of the complement cascade, and is composed of complement factors C5b, C6, C7, C8 and C9 that assemble as a pore on cell membranes. The MAC pore induces ionic fluid shifts leading to cell destruction and ultimate death.
HMR59 works by increasing the production of sCD59 by ocular cells. The sCD59 released from the cells will circulate throughout the eye and penetrate the retina to block MAC deposition and prevent cellular destruction. By blocking MAC, the remainder of the upstream complement cascade is left intact to perform its normal homeostatic roles.
HMR59 will be injected directly into the vitreous cavity in an office setting. Such a procedure is currently performed by all retinal specialists using other medications quite commonly. Using the gene therapy approach offers the opportunity to reduce the number of injections needed over a patient's lifetime as the ocular cells will act as factories to produce sCD59, thus addressing the issue of drug delivery.
In summary:
• The complement pathway is strongly associated with AMD
• Membrane attack complex (MAC) is the final step in the complement pathway
• CD59, a naturally occurring protein, protects retinal cells from damage by MAC
• Hemera Biosciences has developed a gene therapy which produces soluble CD59 (sCD59) that blocks MAC
• A single intravitreal injection allows for long term protection from AMD progression
Hemera Biosciences is currently seeking funding to begin animal toxicology studies to get to a phase 1 study in humans.









1 Comments:
Hi Irv,
Thanks for this Blogg. VERY educational.
sf
www.investorstemcell.com
Post a Comment
<< Home